Lung Diseases
COPD
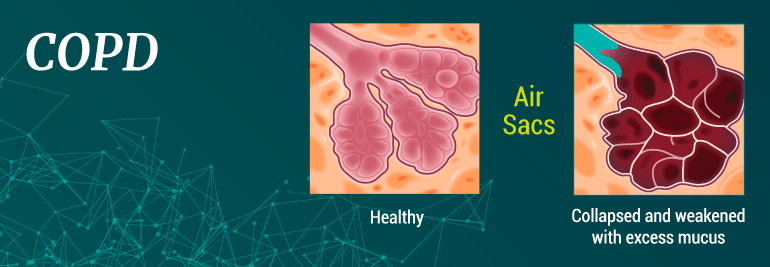
Chronic obstructive pulmonary disease (COPD) is a progressive lung condition — ranging from mild to severe — that is characterized by difficulty breathing and restricted airflow into and out of the lungs. COPD is an umbrella term encompassing chronic bronchitis and/or emphysema.
Emphysema

Chronic Bronchitis

Interstitial Lung Disease

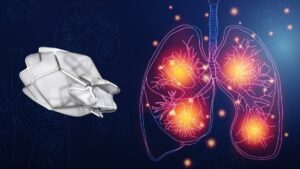
The term “interstitial lung disease” does not describe a single lung condition. It is actually an umbrella term that is used to describe more than 100 different types of pulmonary disorders – many of which are characterized by scarring in the lung’s delicate tissues.
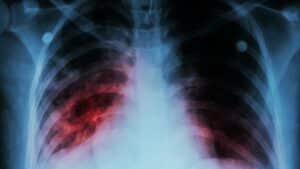
Pulmonary fibrosis

Bronchiectasis

Pneumoconiosis

Stemming from the Greek language for “dusty lungs,” pneumoconiosis is an occupational type of interstitial lung disease caused by the repeated inhalation of mineral dust at job sites. The minerals irritate and inflame the lungs, leaving permanent scars.